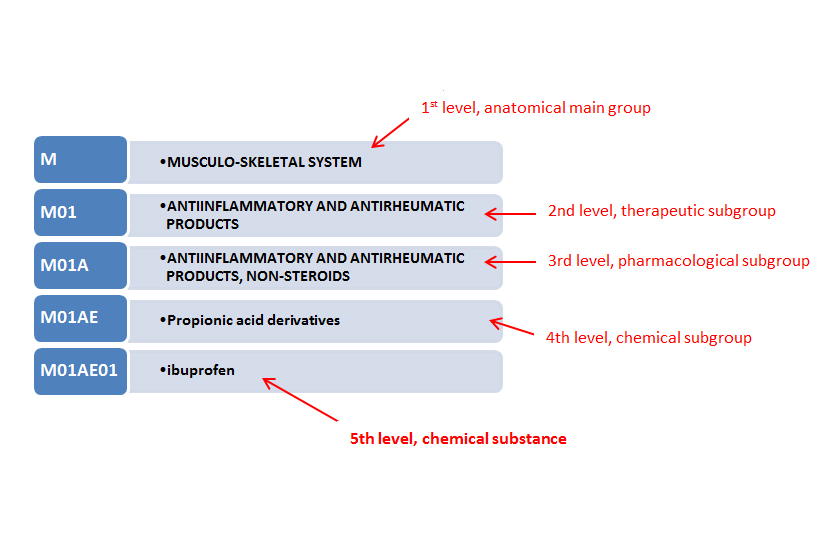
ABOUT THE ATC SYSTEM The Anatomical Therapeutic Chemical (ATC) Classification System is used for the classification of active ingredients of drugs according to the organ or system on which they act and their therapeutic, pharmacological and chemical properties. It is controlled by the World Health Organization Collaborating Centre for Drug Statistics Methodology (WHOCC), and was first published in 1976. This pharmaceutical coding system divides drugs into different groups according to the organ or system on which they act or their therapeutic and chemical characteristics. Each bottom-level ATC code stands for a pharmaceutically used substance, or a combination of substances, in a single indication (or use). This means that one drug can have more than one code: acetylsalicylic acid (aspirin), for example, has A01AD05(WHO) as a drug for local oral treatment, B01AC06 (WHO) as a platelet inhibitor, and N02BA01 (WHO) as an analgesic and antipyretic. On the other hand, several different brands share the same code if they have the same active substance and indications. History In order to measure drug use, it is important to have both a classification system and a unit of measurement. This need was formulated in the late sixties, which still was an infancy period in the field of drug utilization research. Due to an increasing interest in this field, a symposium entitled The Consumption of Drugs took place in Oslo in 1969 organized by the WHO Regional Office for Europe. It was at this symposium The Drug Utilization Research Group (DURG) was established tasked with the development of internationally applicable methods for drug utilization research. Inspired by this interest, a system named the Anatomical Therapeutic Chemical (ATC) classification was developed by the Norwegian Medicinal Depot (NMD). The ATC system is based on the earlier Anatomical Classification System, which is intended as a tool for the pharmaceutical industry to classify pharmaceutical products (as opposed to their active ingredients). This system, confusingly also called ATC, was initiated in 1971 by the European Pharmaceutical Market Research Association (EphMRA) and is being maintained by the EphMRA and the Pharmaceutical Business Intelligence and Research Group (PBIRG). Its codes are organized into four levels. The WHO's system, having five levels, is an extension and modification of the EphMRA's. It was first published in 1976. Classification In this system, drugs are classified into groups at five different levels: First level The first level of the code indicates the anatomical main group and consists of one letter. There are 14 main groups: Code Contents A Alimentary tract and metabolism B Blood and blood forming organs C Cardiovascular system D Dermatologicals G Genito-urinary system and sex hormones H Systemic hormonal preparations, excluding sex hormones and insulins J Antiinfectives for systemic use L Antineoplastic and immunomodulating agents M Musculo-skeletal system N Nervous system P Antiparasitic products, insecticides and repellents R Respiratory system S Sensory organs V Various Example: C Cardiovascular system Second level The second level of the code indicates the therapeutic subgroup and consists of two digits. Example: C03 Diuretics Third level The third level of the code indicates the therapeutic/pharmacological subgroup and consists of one letter. Example: C03C High-ceiling diuretics Fourth level The fourth level of the code indicates the chemical/therapeutic/pharmacological subgroup and consists of one letter. Example: C03CA Sulfonamides Fifth level The fifth level of the code indicates the chemical substance and consists of two digits. Example: C03CA01 Furosemide Other ATC Classification systems ATCvet classification: Anatomical Therapeutic Chemical classification for veterinary medicinal products. It is based on the same main principles as the ATC system for human use; in fact, the ATCvet classification is kept as close to the human system as possible, only introducing the necessary adaptations to make it suitable for the veterinary medicines. ATCvet codes can be created by placing the letter Q in front of the ATC code of most human medications. For example, furosemide for veterinary use has the code QC03CA01. Some codes are used exclusively for veterinary drugs, such as QI Immunologicals, QJ51 Antibacterials for intramammary use or QN05AX90 amperozide. ATC herbal classification: provides a classification of herbal medicines by internationally approved Latin binomial classification and common therapeutic use. Just as the ATC classification, it allows capturing, grouping, and aggregating herbal remedies data at different levels of granularity. Adaptations and updates National issues of the ATC classification, such as the German Anatomisch-therapeutisch-chemische Klassifikation mit Tagesdosen, may include additional codes and DDDs not present in the WHO version. ATC follows guidelines in creating new codes for newly approved drugs. In order to create a new ATC code, an application has to be sent to the WHO. New ATC codes are published twice annually. A formal release of new ATC edition occurs once a year. Inclusion of new entries and principles for classification The WHO Collaborating Centre in Oslo establishes new entries in the ATC classification on requests from the users of the system. Not all substances have an ATC code assigned and the reason might be that no requests has been received for them. The criteria that active ingredients need to fulfill to be included in the ATC system are: Be a new chemical entity or biological proposed for licensing in at least one country (normally a new entity is not included before an application for marketing authorisation is submitted) Existing well defined chemical entities used in a number of countries. Preferably, an INN should be established for the active ingredient, however other official names should be available (e.g. USAN or BAN) herbal medicinal products assessed and approved by regulatory authorities based on dossiers including efficacy, safety, and quality data (such as the procedure in place in the EU) Other medicinal products are considered on a case-by-case basis. Complementary, homeopathic, and herbal traditional medicinal products are in general not included in the ATC system Medicinal products are classified according to the main therapeutic use of the main active ingredient, on the basic principle of only one ATC code for each route of administration, i.e. pharmaceutical forms with similar ingredients and strengths will have the same ATC code. Likewise, immediate and slow release tablets will normally have the same ATC code. Although, a medicinal product may be given more than one ATC code if it is available in two or more strengths or routes of administration with clearly different therapeutic uses. e.g. Finasteride (a specific inhibitor of steroid Type II 5-alfa-reductase (intracellular enzyme that converts testosterone into 5-alfa-dihydrotestosterone) is available in two different strengths: Low strength tablet indicated to treat male pattern baldness D11AX - Other dermatologicals Higher strength tablet used to treat benign prostatic hypertophy (BPH) G04C - Drugs used in BPH In the same way, different pharmaceutical forms developed for various routes of administration (topical or systemic use, for example) are given distinct ATC codes: e.g. The corticosteroid drug prednisolone present in single ingredient products is given different ATC codes depending on the therapeutic use and the corresponding formulation: Enemas and foams Intestinal anti-inflammatory agents A07EA01 Suppositories Anti-hemorrhoidals for topical use C05AA04 Creams, ointments and lotions Dermatological preparations D07AA03 Tablets, injections Corticosteroids for systemic use H02AB06 Nasal sprays, drops Nasal decongestants R01AD02 Eye drops Ophtalmologicals S01BA04 Ear drops Otologicals S02BA03 Finally, it may happen that a drug may be used for two or more equally important indications and they are usually given only one ATC code. In such cases, the main indication is decided based on the available literature and discussed in the WHO International Working Group for Drug Statistics Methodology, where the final classification is decided. Classification of combination products It is interesting to know how medicinal products containing two or more active ingredients are classified in the ATC system. Firstly, combination products are given different ATC codes versus the product with a single component. Even though it may be difficult to decide where a certain combination product should be placed, it is the main therapeutic use what influences how it is classified: e.g. a medicinal product containing an analgesic and a tranquilizer, which is used primarily to ease pain, should be classified as an analgesic. In some ATC groups a ranking is introduced to help in the classification of combination products (e.g. combinations of different antihypertensives or combinations of different analgesics). This ranking shows which drug takes precedence over others when the classification is decided and is detailed in the guidelines for the relevant groups. Specifically, combinations containing two or more active ingredients not belonging to the same 4th level are classified using the 50-series: e.g. Paracetamol ? N02BE01 vs paracetamol, combinations ? N02BE51 Different combination products sharing the same main ingredient are usually given the same ATC code: e.g. Combinations of paracetamol + acetylsalicylic acid and paracetamol + caffeine are classified under the same code N02BE51 paracetamol, combinations All the active ingredients of a combination are given in some ATC 5th levels ? this principle is more commonly used in recent years to give a better identification of the various combinations. e.g.: M01AE02 naproxen M01AE52 naproxen and esomeprazole M01AE56 naproxen and misoprostol Brief insight into the DDDs Even though DDDs are not used in the eHDSI Project, it is worth knowing what they are and their connection with the ATC codes. The DDDs or Defined Daily Doses are the units of measure used in combination with the ATC codes to perform consumption analysis. The DDD is the assumed average maintenance does per day for a drug used for its main indication in adults. As a unit of measure, a DDD should not be confused with the recommended or prescribed daily dose; doses for individual patients, patients groups, other indications different to the main one may differ from the DDD. Only one DDD is assigned to ach ATC code and route of administration for analytical purposes. DDDs provide a fixed unit of measure independent of price, currencies, package size and strength, enabling researchers to assess trends in drug consumption and to perform comparisons between population groups. The basic principles for DDD assignment are: DDDs are only assigned to drugs with an ATC code and will normally not be assigned before a product for the specific substance is approved and marketed in at least one country One DDD is assigned per route of administration within an ATC code: e.g. The DDD for the antibiotic ciprofloxacin given systemically (J01MA02) is 1g when administered orally and 0.5g if administered via parenteral DDDs are not assigned to some groups of products: topical products, vaccines, antineoplastic agents, allergen extracts, contrast media, and others. Drug utilization studies and the ATC/DDD system As explained above, the ATC/DDD system allows standardizing drug grouping and a stable drug utilization metric for comparing drug use between countries, regions, and individual healthcare institutions or settings, and for examining trends in drug use over time. Drug consumption figures are usually presented as number of DDDs/1000 inhabitants/day or, for hospital use, as DDDs per 100 bed days. Sales or prescription data presented as number of DDDs/1000 inhabitants/day may provide a rough estimate of the proportion within a defined area treated daily with certain drugs: a figure of 10 DDDs/1000 inhabitants/day indicates that 1% of the population on average receives that treatment daily. Use and limitations of the ATC Classification system The development and decisions taken around the ATC system are based on its main purpose, i.e. to serve as a tool for presenting drug utilization statistics with the final aim of improving drug use. Consequently, using the system for other purposes may not be appropriate. For its intended use, comparing drug utilization at international level for instance, it is important that the data retrieved are comparable. For that, the ATC code and DDD should be linked to each medicinal product at package level; sometimes it has been acknowledged by WHO that the necessary skilled staff has not been allocated to this task. An additional issue has been that some users were not aware of the dynamic nature of the classification and that the annual updates may require the subsequent update of the national lists. Finally, there will always exist medicinal products - either single or combination formulations- for which ATC codes or DDDs are not available. Finally, as already mentioned, the ATC classification does not reflect recommended therapeutic use, neither does it imply any judgement about the efficacy of drugs or groups of drugs. References Guidelines for ATC classification and DDD assignment. 2013. 16th edition. WHO Collaborating Centre for Drug Statistics Methodology Norwegian Institute of Public Health. https://en.wikipedia.org/wiki/Anatomical_Therapeutic_Chemical_Classification_System https://ec.europa.eu/cefdigital/wiki/pages/viewpage.action?pageId=52609352 Marit Rønning. Coding and classification in drug statistics – From national to global application // Norwegian Journal of Epidemiology 2001; 11 (1): 37-40.
If this page is in your subscriptions, then it will be removed. You will not see this page. If you want to unblock a user, go to the settings, the list of blocked users and click unblock